 
The vales are put together in a table and those values can be used to calculate the standard molar. 1 The Standard Molar Entropies of Selected Substances at 298. Standard molar entropies of many substances are known. SE measures EEG activity up to a frequency of 32 Hz. Earlier the value of 185.3 J/molK was calculated from experimental data Giauque W.F., 1931. State entropy (SE) and response entropy (RE) values are generated. The calorimetric value is significantly higher than the statistically calculated entropy, 186.26 J/molK, which remains the best value for use in thermodynamic calculations Vogt G.J., 1976, Friend D.G., 1989, Gurvich, Veyts, et al., 1989. The list of property identifiers needed in the calling arguments and instructions are available in the Thermophysical Function help. A table like this can be used in much the same way as a table of standard enthalpies of formation in order to find the entropy change Sm for a reaction occurring at standard pressure and at 298 K. The GE Entropy TM Module (it was developed by Datex-Ohmedam, now part of GE Healthcare) measures these changes by quantifying the irregularity of the EEG signals 6. The JANAF table reference for entropy is based on the Third Law of Thermodynamics which references the entropy of all pure crystalline substances to zero at absolute zero temperature. However, all ideal gas substances (which have a chemical symbol name, e.g., N2, CO2, CH4) have enthalpy values corresponding to JANAF table references. Thermochemical data for over 7000 organic and small inorganic compounds: Enthalpy of formation Enthalpy of combustion Heat capacity Entropy Phase transition. 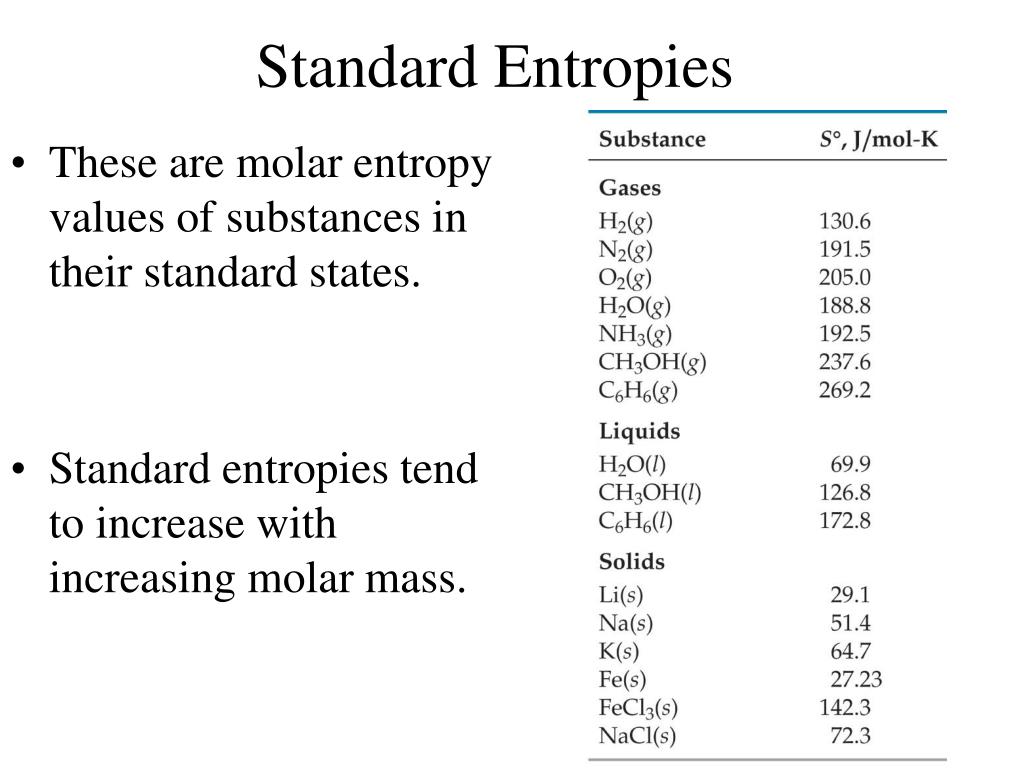
The reference state upon which the value of enthalpy is based varies with the substance. Temperature must be the only argument, in addition to the substance name. The specific entropy of incompressible substances is a function of only temperature. Note also that for substance AirH2O (psychrometrics), the specific entropy returned by this function is the entropy of the air and water vapor mixture per unit mass of dry air. Specific entropy values are obtained from these sources in the same way that any of the other specific properties are obtained. The remaining two can be any of the following: temperature (T), enthalpy (H), internal energy (U), relative humidity (R), humidity ratio (W), wetbulb (B), or dewpoint (D). The tables and charts in Thermodynamic Tables to accompany Modern Engineering Thermodynamicslist specific entropy along with the specific properties v, u, and h. One of these arguments must be total pressure (P). For all pure substances, the entropy function always requires two arguments, in addition to the substance name.įor AirH2O, three arguments are required. The value and units of the returned value depends on the Unit System setting. ENTROPY returns the specific entropy of a specified substance. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
